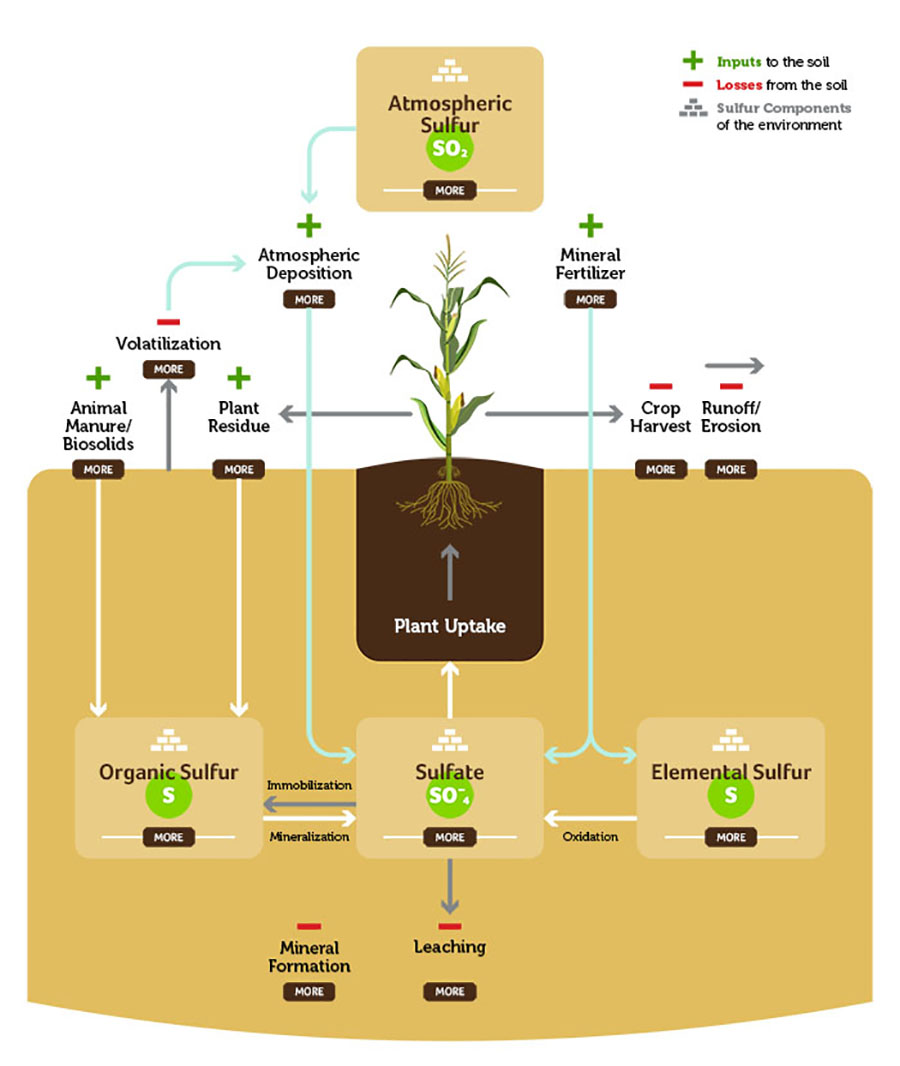
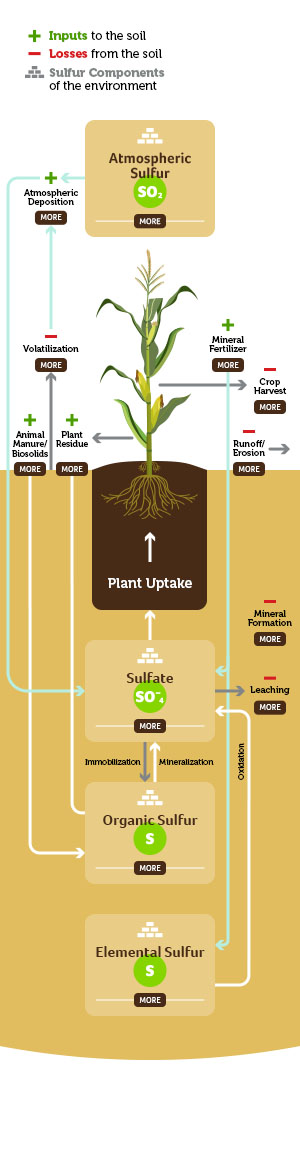
Atmospheric Sulfur
Sulfur is distributed into the atmosphere from burning sulfur containing fossil fuels, volcanic emissions, and other natural processes. As a gas in the atmosphere it exists as sulfur dioxide gas (SO2). Sulfuric acid and sulfate are deposited back on the earth through precipitation. Sulfur deposition through precipitation has decreased steadily since the mid-’80s.
Atmospheric Deposition +
Atmospheric deposition is commonly known as dust settling onto the soil surface. This settling of dust can happen from several things including mines, rock erosion and runoff from rivers and streams.
Mineral Fertilizer +
Depending on the crop being grown and the supply of sulfur from the soil, producers can supplement sulfur nutrition via fertilization. Fertilizer sources can either be in salt forms (ammonium sulfate, calcium sulfate, potassium sulfate, etc.) or as elemental sulfur.
Volatilization –
Sulfur does exist in various gaseous forms that can be lost from the soil system via volatilization. The losses of sulfur in most agricultural soils is quite small (< 0.05 percent of total sulfur in the soil), and not considered a significant loss pathway.
Animal Manure/Biosolids +
Animal manure/biosolids are good sources of sulfur within the soil. The quantity of sulfur in manure depends on the animal species, age and diet, and bedding material. Manure/biosolids supply sulfur in the organic form and thus must be mineralized to provide sulfate to a growing crop.
Plant Residue +
The unharvested plant material returns sulfur in an organic form back to the soil. Sulfur in plant residues becomes available as the residues are decomposed by soil organisms.
Crop Harvest –
This is obviously the desired sulfur destination within a crop production system. Crops take up sulfate exclusively.
Runoff/Erosion –
Surface losses of sulfur can occur when sulfate is near the surface during high intensity precipitation events that result in water runoff. Surface losses of sulfur can also occur when soil sediment (which contains sulfur – organic form, sulfate form, mineral forms) is lost through erosion.
Organic Sulfur
Organic sulfur becomes available when soil organic matter is mineralized to sulfate. Organic matter generally has a makeup of carbon:nitrogen:sulfur in a ratio of 120:10:1.4. Organic sulfur mineralizes (releases sulfate) when the carbon:sulfur ratio is less than 200:1. Sulfate can be immobilized (move from available sulfate to organic sulfur) when the carbon:sulfur ratio is greater than 400:1.
Sulfate (SO-4)
Negatively charged sulfate ions (anions) in soil do not react with predominantly negatively charged soil particles. As a result, sulfate may leach out of the root zone with excess rainfall or accumulate at the soil surface in dry conditions. Sulfate sulfur is the form taken up by plants.
Elemental Sulfur
Elemental sulfur is not a form that is immediately plant available. Elemental sulfur requires biological oxidation to convert it into sulfate. There are several sulfur oxidizing bacteria that can facilitate this reaction.
Leaching –
The sulfate form of sulfur (SO42-) is soluble and can leach when excess water moves through the soil. With the help of microorganisms and bacteria in the soil, sulfur transforms into sulfates. In coarse-textured soils, leaching can be a major loss, but less so in finer-textured soils.
Mineral Formation –
Sulfur can form relatively insoluble minerals in soils, but those reactions occur under very specific conditions. As an example, in arid soils, sulfate can precipitate into soluble and insoluble salts. In high pH soils, sulfate can react with free calcium carbonate (lime) to form lime/gypsum (calcium sulfate) complexes. Finally, under long-term anaerobic conditions, sulfides (coastal regions/paddy production systems) can accumulate and complex with iron.